Community
osapeers.org

Live in
days
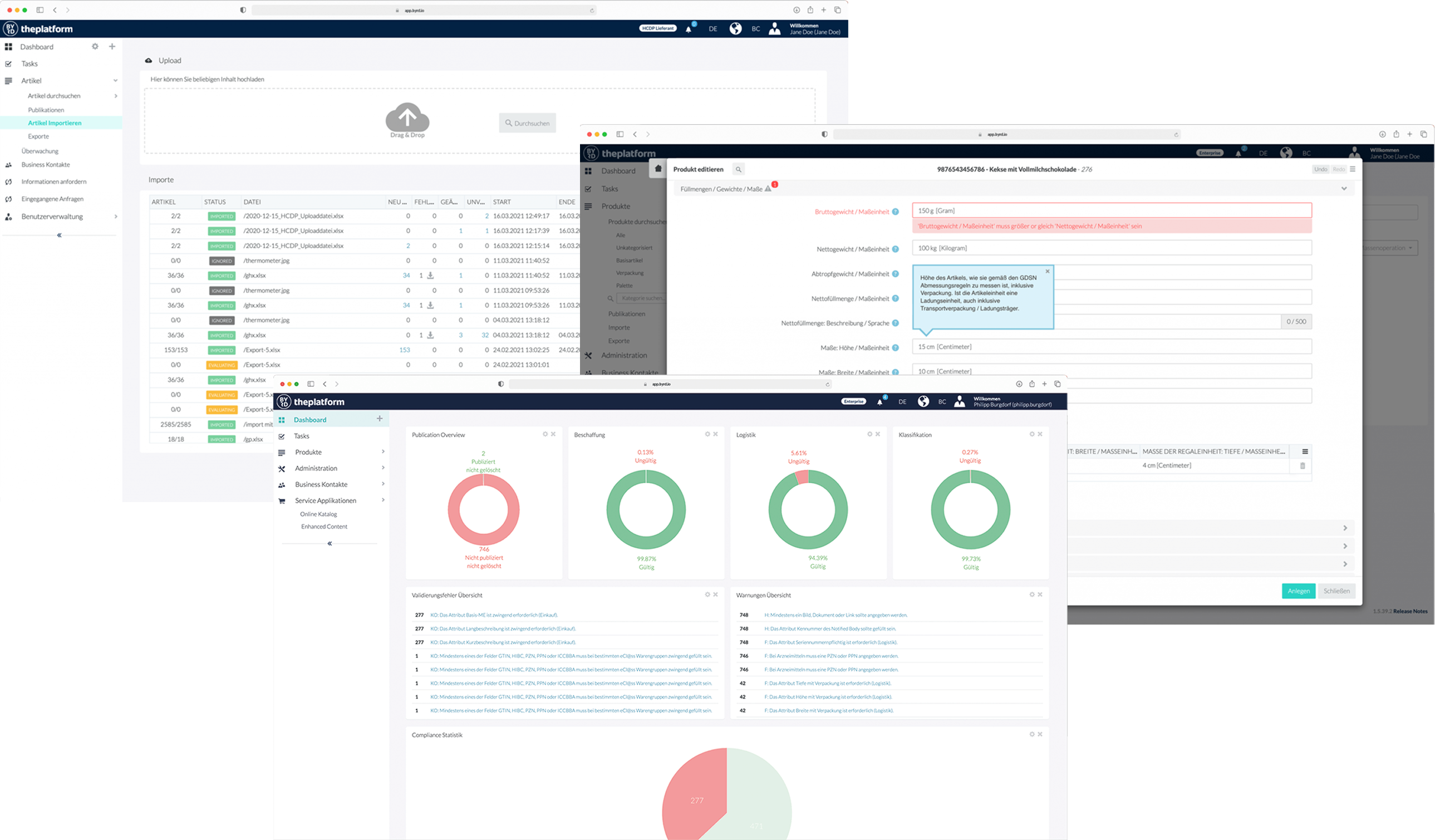
Complex Data Submission Processes
Manage your medical device data efficiently while ensuring compliance, accuracy, and audit readiness across global regulatory registries with the osapiens HUB for Medical Devices:
✓ Centralize Your Data: Collect all product information in one place, keeping internal teams, distributors, and hospitals aligned.
✓ Validate Automatically: Ensure your UDI and product data meet regulatory requirements before submission, minimizing errors and compliance risk.
✓ Syndicate Globally: Submit data efficiently to EUDAMED, GUDID, and other regulatory authorities worldwide.
✓ Accelerate Time-to-Market: Reduce friction in data exchange and bring new products to market faster.
✓ Audit-Ready & Traceable: Maintain a fully auditable history and ensure compliance with QMS and global standards.
EUDAMED





Operate in a validated, QMS-compliant environment with the osapiens HUB for Medical Devices to exchange UDI data securely and compliantly between manufacturers, regulators, and trading partners.
Our in-house experts, who co-chair the EUDAMED IT Expert Group at MedTech Europe and contribute to the GS1 Global Healthcare Leadership Team, ensure your data management aligns with the latest regulations and industry best practices.


EUDAMED will become mandatory on May 28, 2026. This is your chance to understand exactly what you need to do now to stay compliant and avoid costly delays.
Want to stay uptodate? Watch the free recording of our latest expert-led webinar!





Resource Center